:::
- 首頁
- /
- 年度
- /
- 2021
- /
- 精準健康(醫材&防疫科技)
- /
- 子宮內膜癌安蓓甲基化基因檢測
| 技術名稱 | 子宮內膜癌安蓓甲基化基因檢測 | ||
|---|---|---|---|
| 計畫單位 | 臺北醫學大學 | ||
| 計畫主持人 | 賴鴻政 | ||
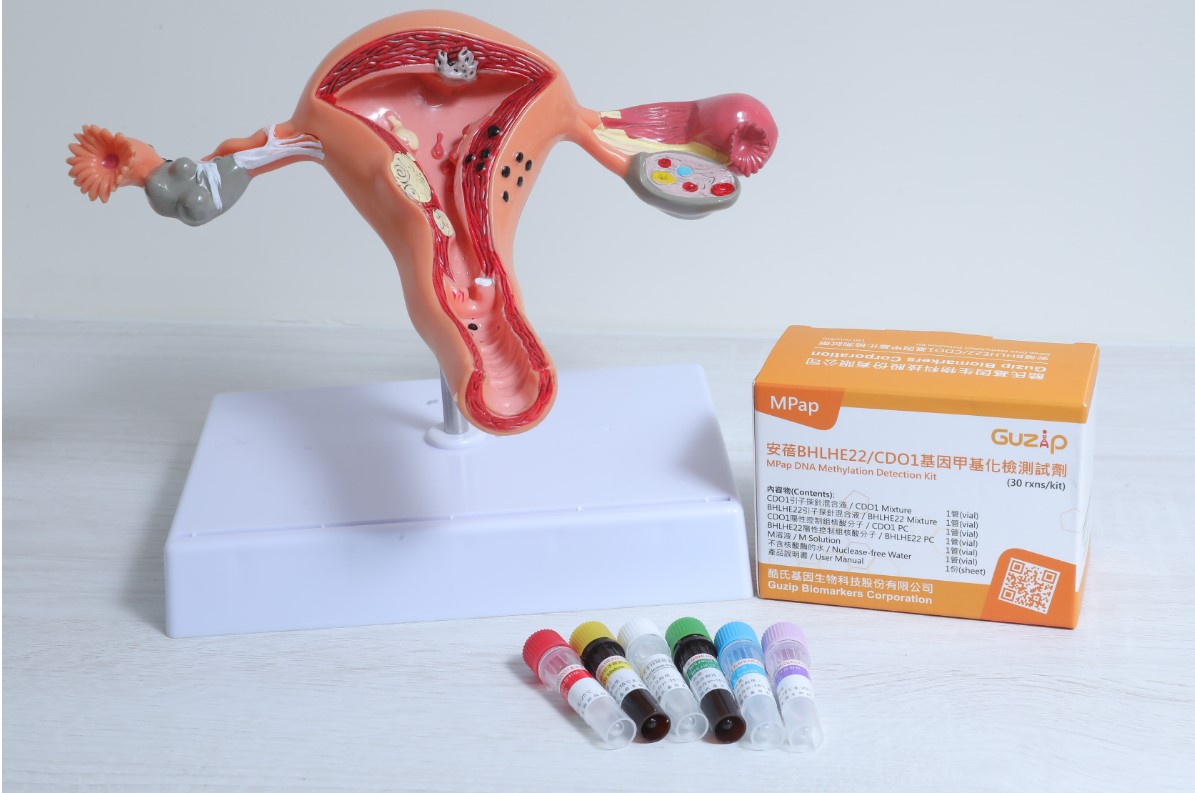
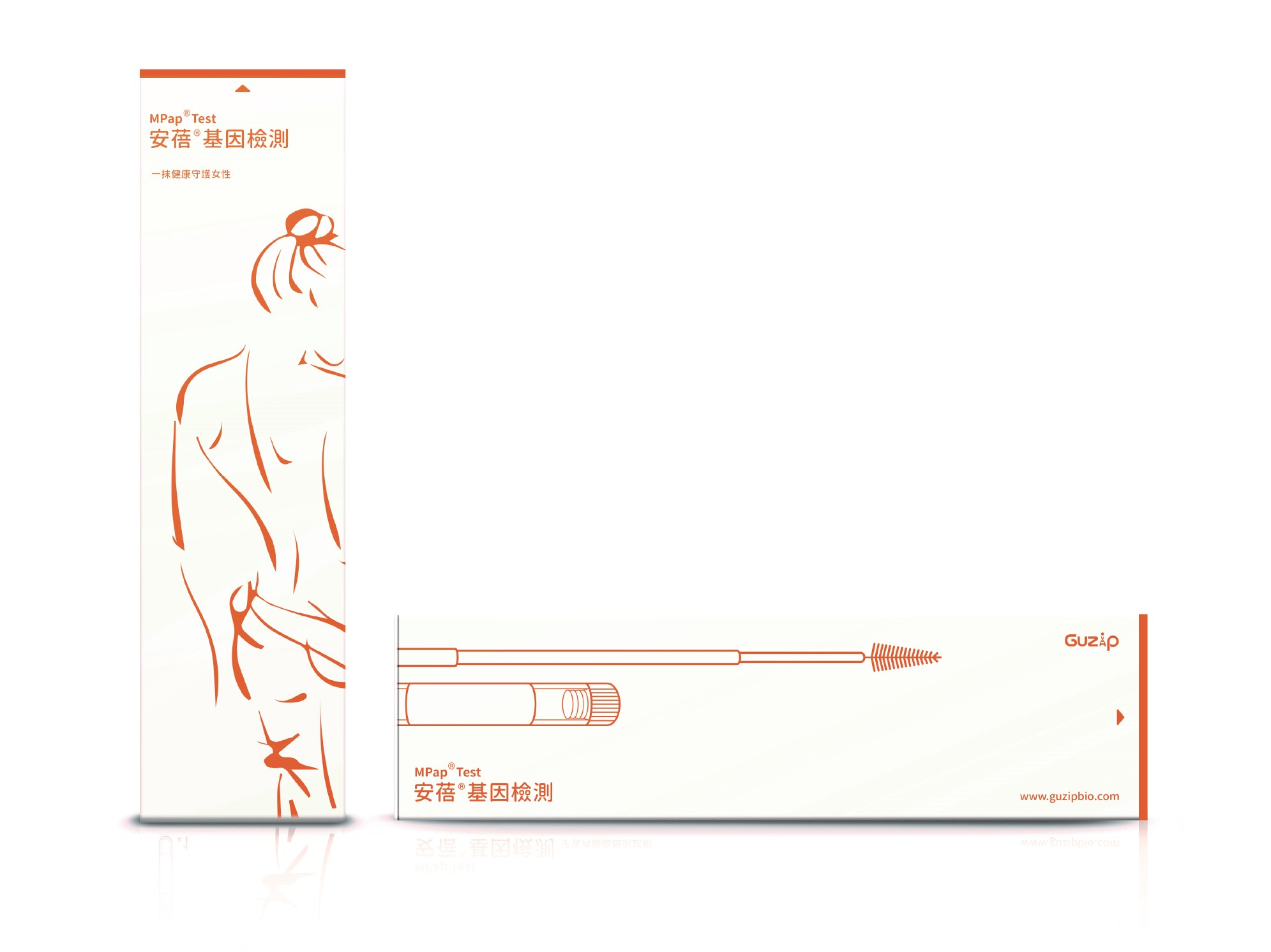
| 技術簡介 | 安蓓基因檢測是一種子宮內膜癌的體外診斷試劑,針對40歲以上子宮異常出血的婦女,利用子宮頸抹片的檢體,偵測BHLHE22及CDO1兩個基因的甲基化狀態,評估子宮內膜癌的風險,提供醫師是否進行侵入性檢查的重要依據,本技術可大幅減少侵入性檢查的必要性,同時提高子宮內膜癌篩檢的可行性,為領先全球的技術。 |
||
| 技術影片 | |||
| 科學突破性 | 子宮內膜癌,到目前為止,並沒有分子診斷工具。安蓓甲基化基因檢測,為目前世界上首先完成臨床驗證之分子診斷試劑。經台灣五大醫學中心的臨床試驗,在五百多位受試者的檢體得到靈敏度為91、特異度為73、AUC為0.9。此結果與目前所使用的陰道超音波檢查進行平行比較,都遠優於超音波。 |
||
| 產業應用性 | 安蓓甲基化基因檢測利用傳統抹片的取樣法,早期偵測子宮內膜癌,為市場上第一個針對子宮內膜癌的分子標記,準確性高,可提供醫師評估侵入性組織採檢的重要依據,減少侵入性檢查的風險,並節省醫療資源。 |
||
| 媒合需求 | 天使投資人、策略合作夥伴 |
||
| 關鍵字 | 甲基化基因 子宮內膜癌 體外診斷試劑 子宮頸抹片 子宮異常出血 安蓓 | ||
- 聯絡人
- 黃珮瑩
- 電子信箱
- gyntsgh2@gmail.com