:::
- 首頁
- /
- 年度
- /
- 2021
- /
- 精準健康(醫材&防疫科技)
- /
- 泌尿道上皮癌微型 RNA 篩檢套組
| 技術名稱 | 泌尿道上皮癌微型 RNA 篩檢套組 | ||
|---|---|---|---|
| 計畫單位 | 國立中央大學 | ||
| 計畫主持人 | 馬念涵 | ||
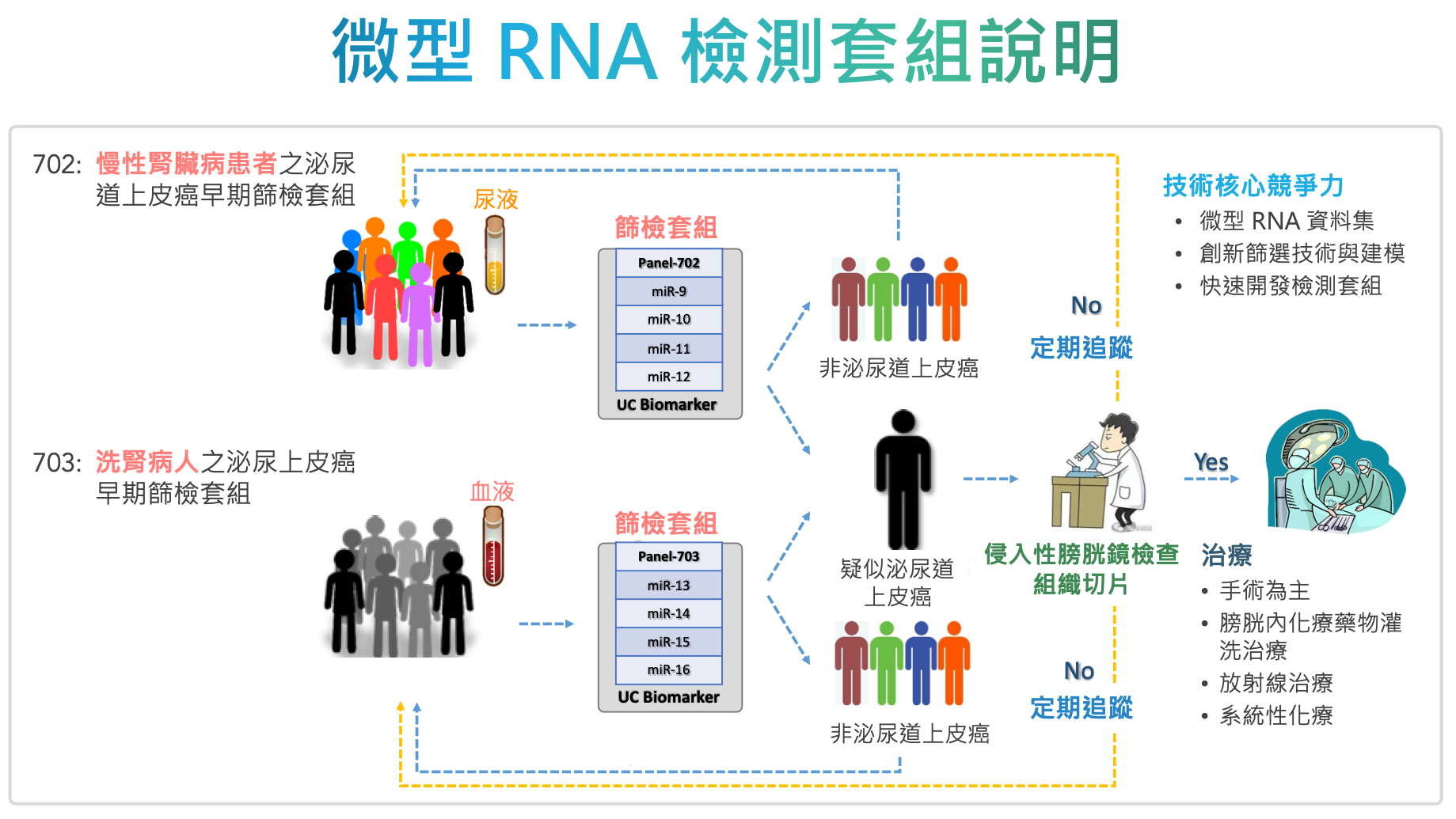
| 技術簡介 | 專為泌尿道上皮癌高風險族群開發檢測套組,以高度專一性miRNA組合作為疾病標記,具有極低侵入性和高敏感度之優勢與競爭力。慢性腎臟病患者使用尿液,洗腎病人使用血液進行檢測,如結果疑似罹癌,再進一步接受高侵入性泌尿道內視鏡及切片檢查,如經確診可及早治療,提升治癒率及存活率。如結果為陰性,則每年定期追蹤。 |
||
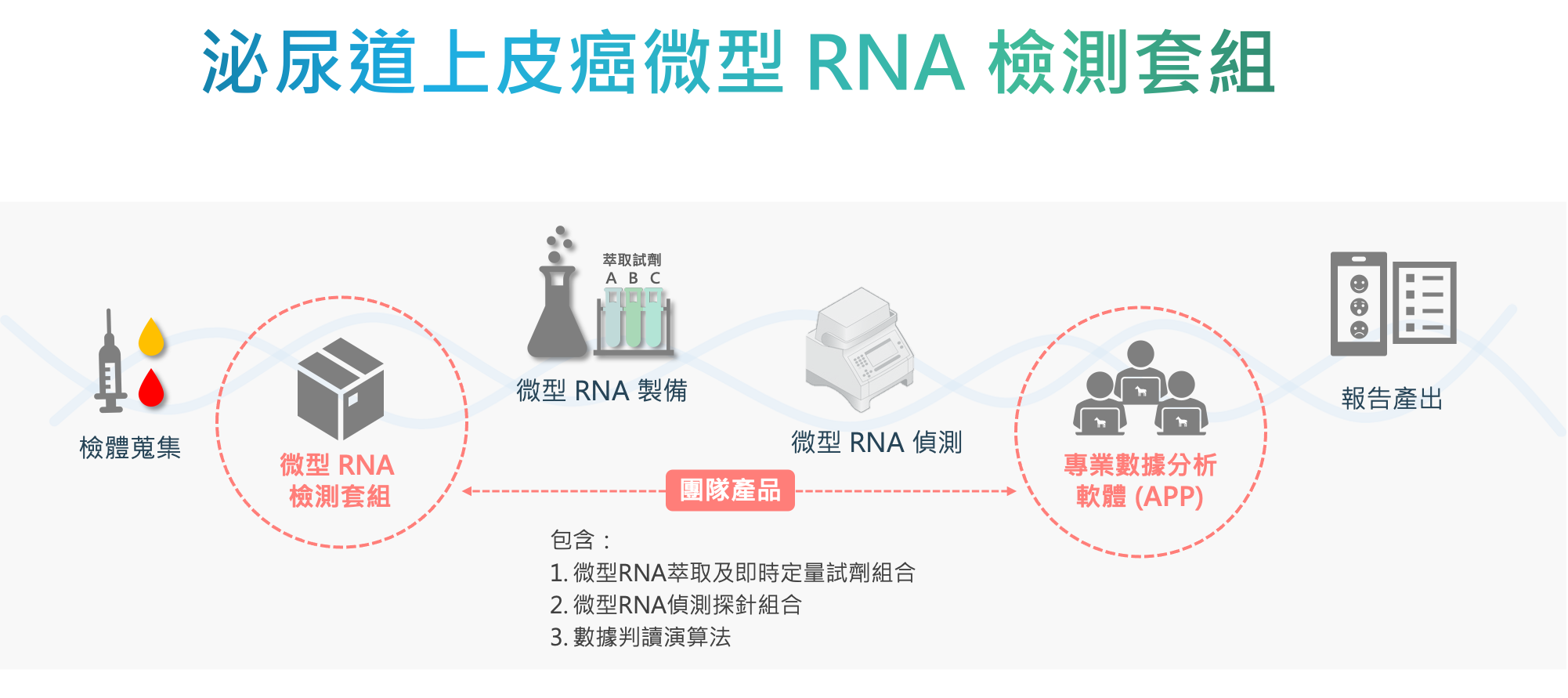
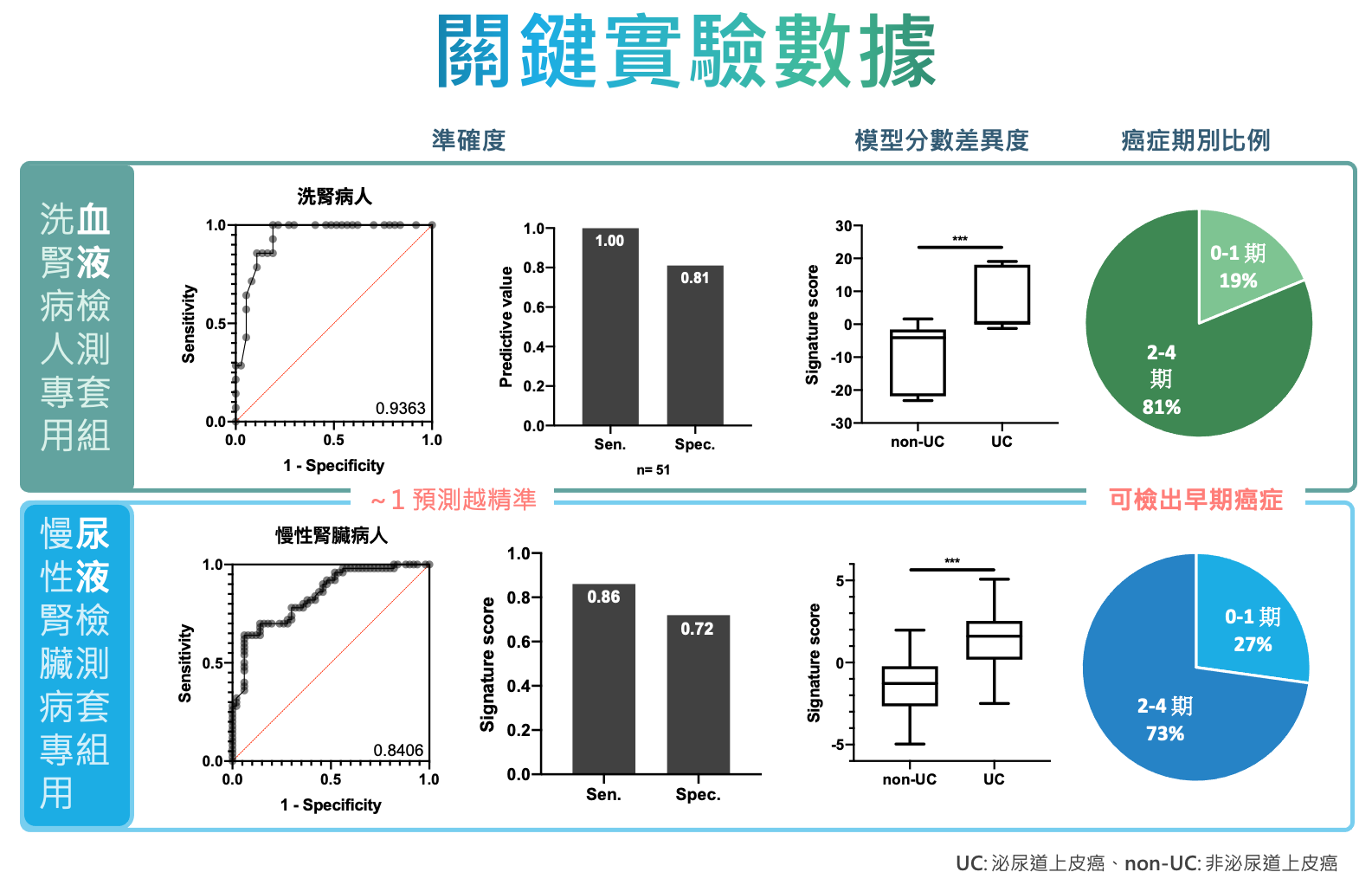
| 科學突破性 | 團隊開發符合高風險族群特定需求的檢測方法,挑選具有泌尿道上皮癌組織特異性的miRNA組合作為疾病標記,搭配市面上常見之qRT-PCR設備,測量這些miRNA的組合表現量,透過miRNA比值評估相對表現量,不僅能穩定地偵測體液中的微量因子,同時滿足高度敏感性和專一性,以及極低侵入性之要求。 |
||
| 產業應用性 | 團隊的泌尿道上皮癌微型 RNA 篩檢套組專為高風險族群開發,在不影響現有泌尿道上皮癌篩檢與追蹤的臨床流程下,提供極低侵入性、高靈敏度及具病患福祉的檢測工具。有別現有篩檢方法上的限制與不適感,藉由降低病患使用侵入式檢查上的風險與心理壓力,提高了病患做檢測或定期檢測的意願,進而提升癌症的治癒率與存活率。 |
||
| 媒合需求 | 天使投資人、策略合作夥伴 |
||
| 關鍵字 | 微型RNA 泌尿道上皮癌 慢性腎臟病 洗腎 疾病標誌 篩檢套組 分子診斷 即時定量PCR 精準醫療 miRNA檢測開發平台 | ||
- 聯絡人
- 鍾沛容
- 電子信箱
- charlie850729@gmail.com
其他人也看了