| 技術名稱 | 新冠肺炎的診斷與治療 | ||
|---|---|---|---|
| 計畫單位 | 中央研究院 | ||
| 計畫主持人 | 吳漢忠 | ||
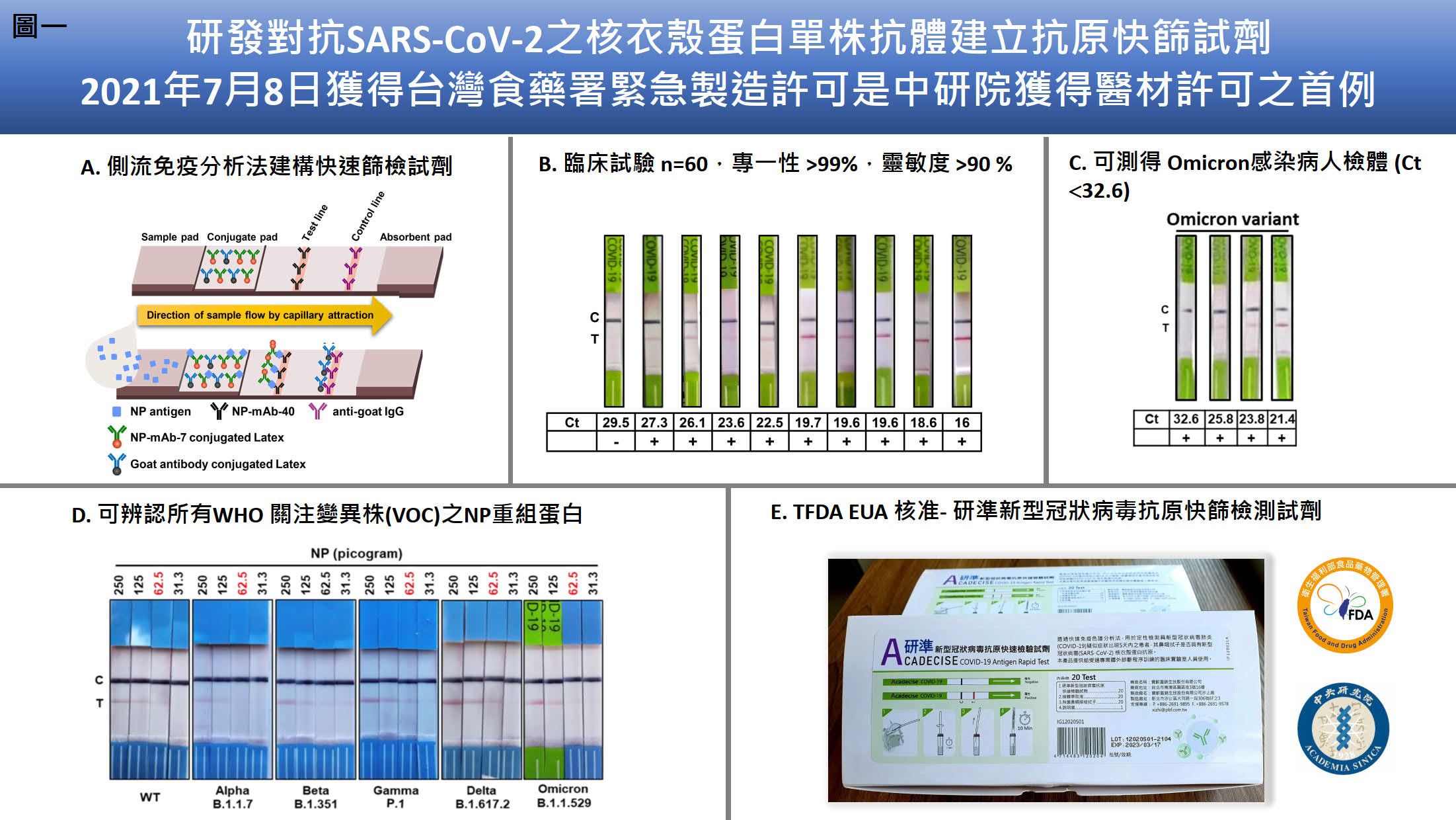
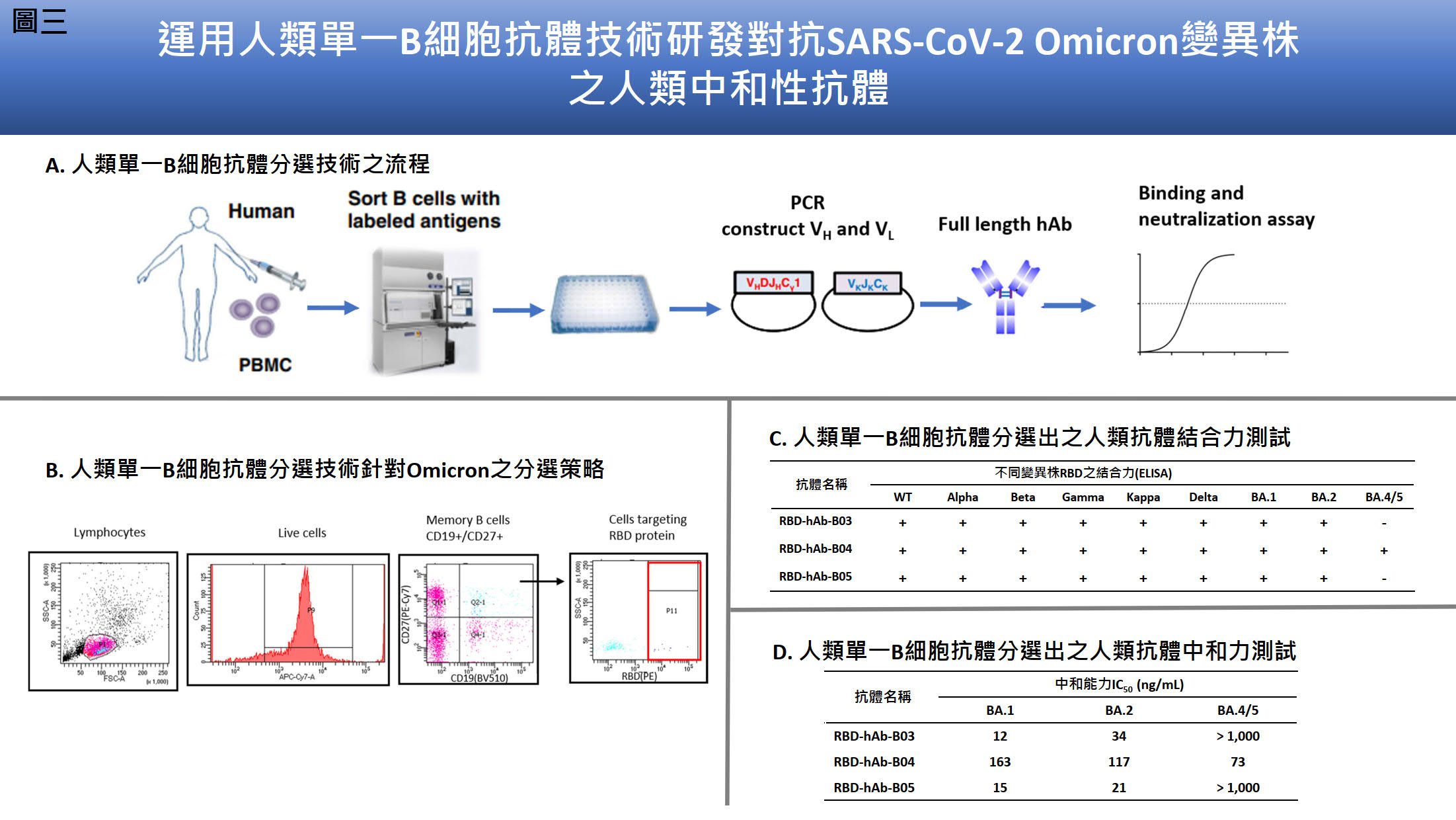
| 技術簡介 | 單株抗體技術生產對抗核衣殼蛋白並運用於研發抗原快篩試劑-研準,具高度專一與靈敏度獲得台灣食藥署的緊急製造核准。為了克服新冠病毒Omicron的免疫逃逸,我們使用mRNA-脂質奈米粒子免疫方法、抗體工程與單一B 細胞抗體平台,研發人源化與人類抗體群能廣泛中和各種變異株包括Omicron BA.4/5。 |
||
| 科學突破性 | 研準與美國FDA認證類似快篩產品之最低偵測極限相比,排名第六名具有國際競爭力;臨床顯示研準能測得病毒量極低(Ct ≤32.6)之Omicron陽性之鼻咽檢體展現高度靈敏度。mRNA- LNP合成快速,節省傳統抗原蛋白製備的時程,並在小鼠體內表現正確構型抗原蛋白,此免疫技術是病毒抗體研發的重要進展。 |
||
| 產業應用性 | 研準新冠病毒抗原快篩試劑已通過臨床試驗,獲得台灣食藥署的緊急製造核准,是中研院獲得醫材許可之首例,待製造經銷合約簽署後,即可上市投入防疫檢測任務。廣效性中和抗體除了運用於治療,亦可使用於預防,幫助免疫低下族群快速大量提升中和抗體濃度,預防新冠肺炎中重症的發生。 |
||
| 關鍵字 | 新冠肺炎 新冠病毒 中和性抗體 抗原快篩試劑 人源化抗體 mRNA-脂質微粒 | ||
- 聯絡人
- 呂瑞旻
- 電子信箱
- reminlu@gate.sinica.edu.tw