| 技術名稱 | 治療多重系統退化症的新希望 | ||
|---|---|---|---|
| 計畫單位 | 國立臺灣大學 | ||
| 計畫主持人 | 賴文崧 | ||
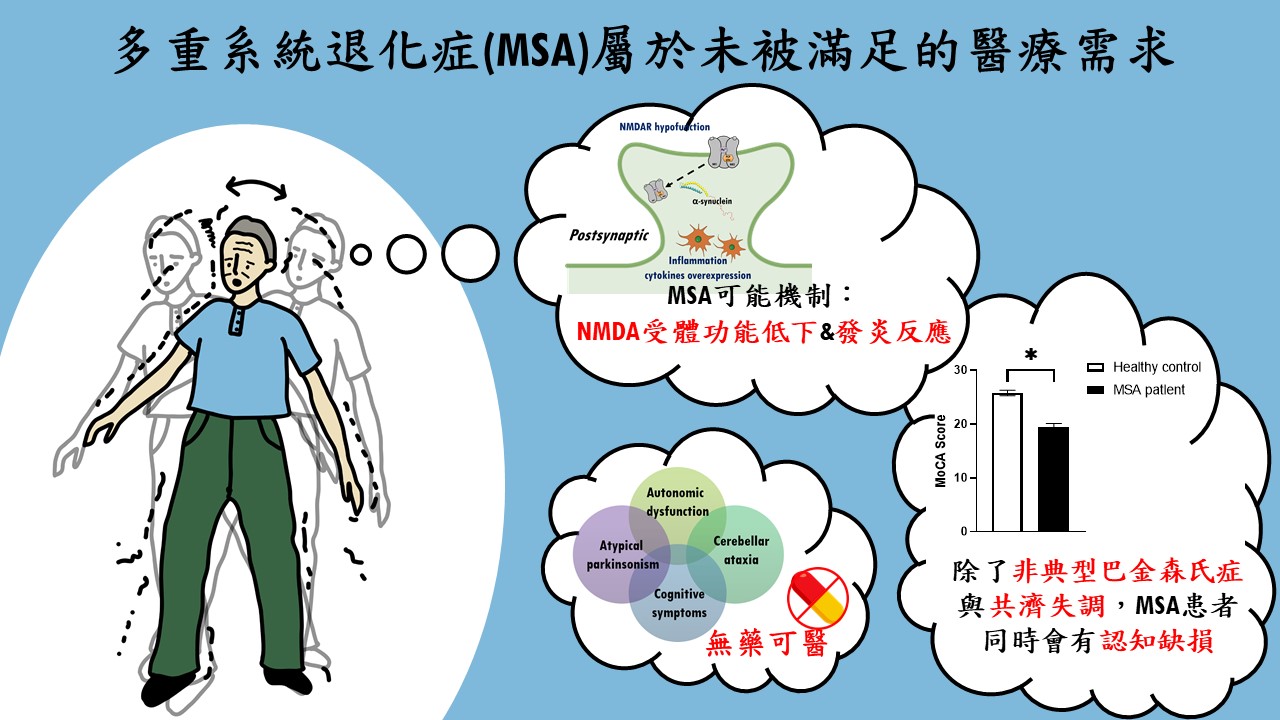
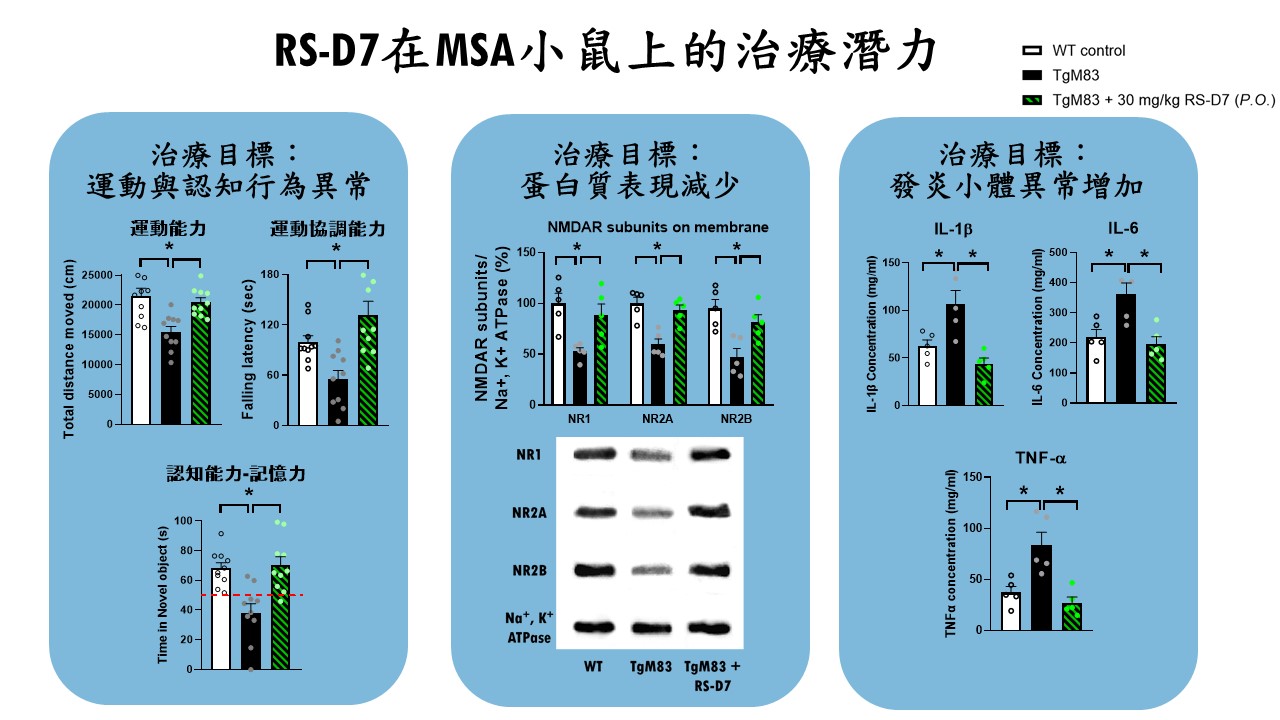
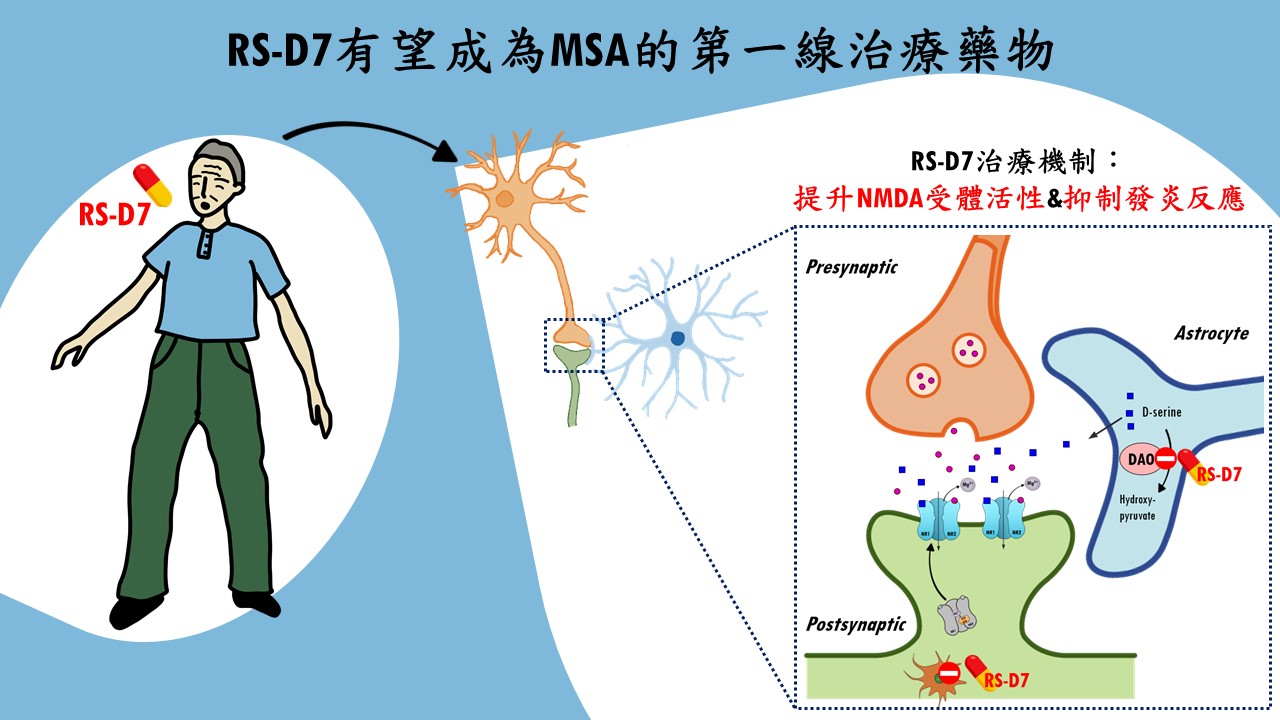
| 技術簡介 | MSA是一種成年發病且致命的神經退化疾病,臨床上表現非典型巴金森症,患者有動作障礙及自主神經失調症狀,發病至死亡平均9年,缺乏有效的治療藥物。團隊針對NMDA受體研發新成分藥物RS-D7有很高的安全性,能有效地改善MSA小鼠相關的動作及認知功能,已取得多國專利及完成技轉,可望為MSA治療開啟新希望。 |
||
| 科學突破性 | MSA為迫切未被滿足的醫療需求。團隊突破傳統窠臼從DAO抑制劑著手,以臨床前動物模型搭配行為表現型技術以及神經機制探討,證實RS-D7可透過調節NMDA受體以及抑制發炎反應,有效改善MSA小鼠的運動協調功能以及認知缺損。RS-D7的研發成果為迄今仍無藥可治的多重系統退化症帶來新希望。 |
||
| 產業應用性 | MSA迄今仍無藥可治,預估其藥物市場每年至少1.5億美元。RS-D7可望成為MSA第一線治療藥物,可有效改善相關運動與認知缺損問題,RS-D7陸續獲得國家新創獎與未來科技獎等肯定,擁有多國專利,並通過FDA孤兒藥資格,可使用快速審查加速發展。已順利技轉授權,即將進行人體臨床試驗,極具開發投資性。 |
||
| 關鍵字 | 多重系統退化症 共濟/運動失調 認知缺損 新藥研發 NMDA受體 d-胺基酸氧化脢抑制劑 罕病用藥 未被滿足的醫療需求 專利 | ||
- 聯絡人
- 羅達中
- 電子信箱
- D06227102@ntu.edu.tw
其他人也看了