| 技術名稱 | 婦科疾病精準體外快篩系統 | ||
|---|---|---|---|
| 計畫單位 | 國立中山大學 | ||
| 計畫主持人 | 許晉銓 | ||
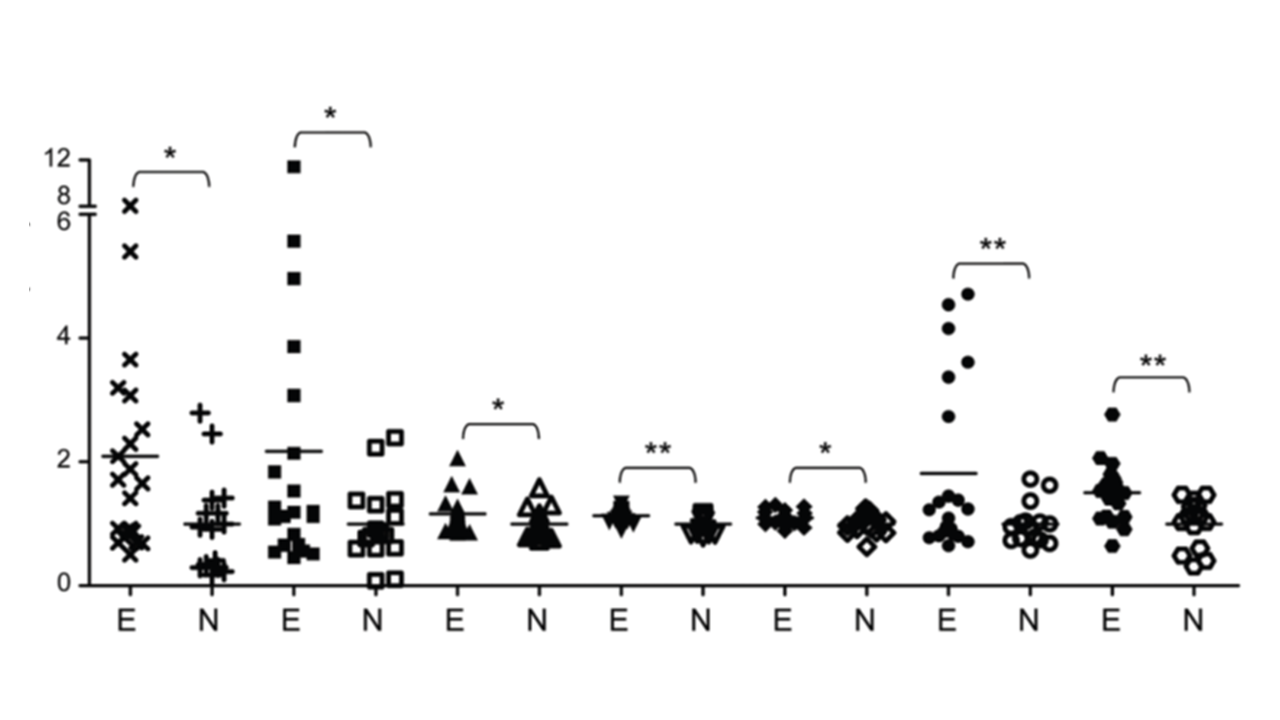
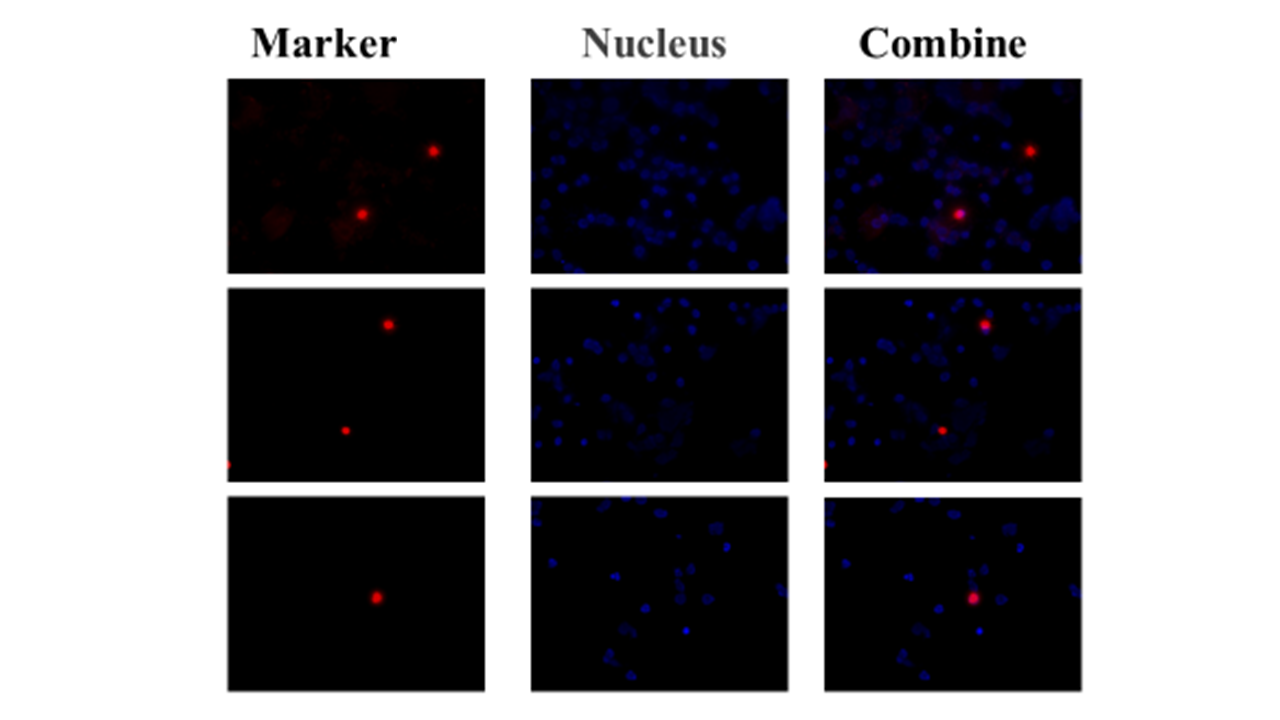
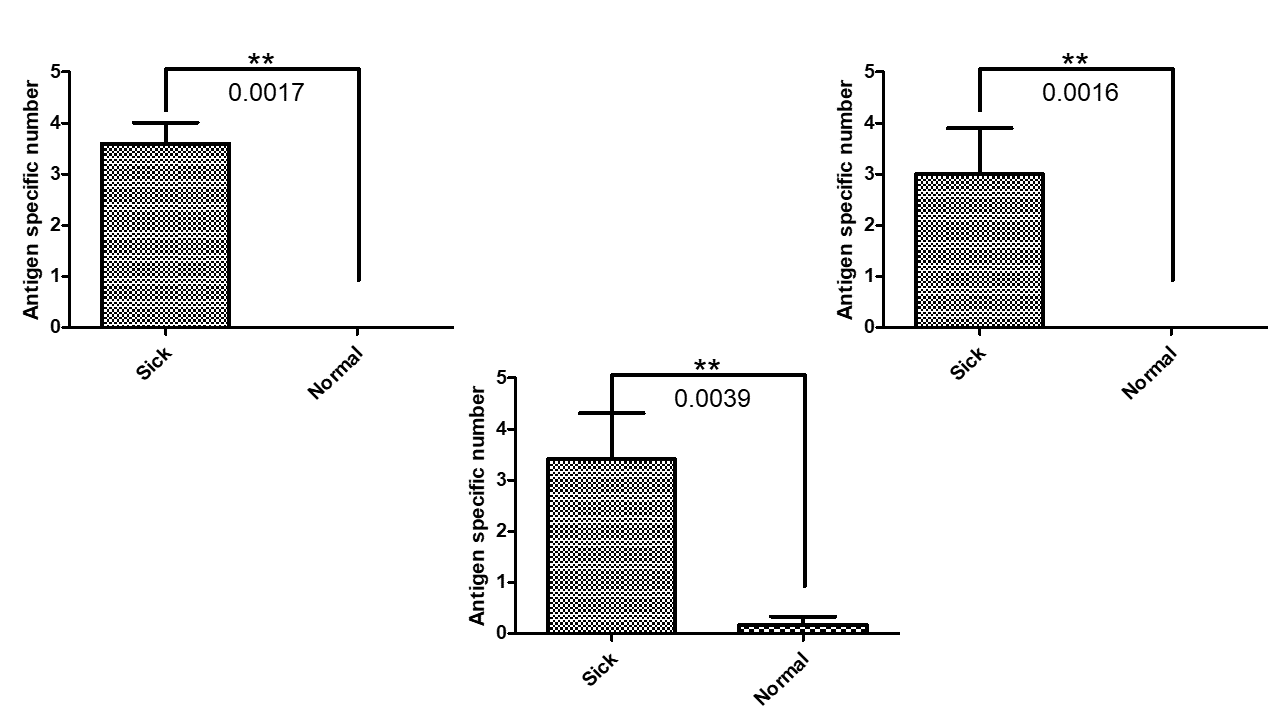
| 技術簡介 | 子宮內膜異位症長久以來為一種令婦科苦惱之疾病,全球平均育齡女性發病率約為 10%,此疾病會對婦女日常生活造成嚴重影響,包含崩潰性劇烈腹部疼痛、器官大量出血、性行為困難、導致不孕症甚至引發高風險的癌症惡化可能性(比常人高出4.2倍卵巢癌發生率;1.3倍乳癌發生率;1.4倍血癌發生率)。目前傳統檢方法測雖然嚴謹並行之有年,但相對複雜耗時,且通常只有中大型醫療機構才擁有這些儀器設備,造成民眾至這些醫療機構就診費時又耗體力,著實不便,同時很多民眾因為對醫院的排斥心理,也都是在身體不適到無法忍受時才願意就醫,但通常這時候疾病已經進入中後期惡化階段了,所以若是單純只仰賴中大型醫療院所,在實際執行層面上其實很難做到疾病早期篩檢。我們從分子醫學的角度切入,領先全球發現核醣體的核型變異與子宮內膜異位症之惡化及癌化有密切的關聯性,當大量表現變異核醣體的RNA時會活化許多參與rRNA修飾與蛋白生成之重要基因,而在子宮內膜組織內的核醣體生合成效率確實相較於正常組織來的活化,且會隨著疾病惡化而跟著加重表現,因此我們透過與中國醫藥大學附設醫院婦產部共同合作完成超過60組的臨床試驗,開發準確的檢測標的組合,同時具備精準、快速且方便三項優點,我們的技術可準確偵測使用者的子宮內膜異位症從早期到晚期不同階段的資訊,幫助做到疾病的早期預防,同時因具有居家執行的方便性,可以大大增加疾病檢測的速度及人數範圍,幫助彌補醫療機構不易執行早期檢測的缺口,避免患者在疾病中後期才確診所帶來的風險及龐大醫療費用,真正做到落實預防醫學的理念,並也藉此希望能夠降低未來相關癌症的病發風險,帶給全球女性及其家人更安心的未來。 |
||
| 科學突破性 | 目前對於子宮內膜異位症的診斷方法雖然有其權威性,但因檢測項目太多,耗費大量時間及體力,實際執行上不方便做早期篩檢。而我們研發之體外檢測試劑則同時具備精準、快速等優勢,且操作的方便性省去了過多的體力及時間消耗,因此能夠做到大範圍快篩,且在與Ca-125及競爭對手的比較上,我們的精準度及穩定性表現更優異 |
||
| 產業應用性 | 全球DTC的醫療市場預估在2年後將成長到9億美金,而美國每年有超過60萬的婦女執行子宮切除手術,但若能夠於早期發現,有機會單靠藥物治癒,費用為手術的千分之二。我們的檢測服務不僅能夠做到早期快篩,同時因居家操作便利性,能補足醫院難以施行早期診斷,幫助患者免除手術鉅額費用且挽救子宮切除帶來的嚴重人生影響 |
||
| 關鍵字 | 體外檢測試劑 精準醫學 預防醫學 婦科疾病 | ||
- 聯絡人
- 曾俊誠
- 電子信箱
- n1255bbb@gmail.com
其他人也看了